第3回 バイオフォーラム2011
最先端の生命科学研究に触れてみませんか
第3回
Evolution of questions about protein import channel
The chloroplast is an indispensable organelle of photosynthetic eukaryotes. Its biogenesis depends on binary fission and protein import. Toc75 functions as a protein import channel in the outer envelope, and is essential for plant viability from the embryonic stage. Toc75 is nuclear-encoded and synthesized in the cytoplasm with a cleavable targeting sequence in its N-terminus. Despite its importance, however, the mechanisms of envelope-targeting and complete maturation of Toc75 remained largely elusive. Furthermore, although the prokaryotic origin of Toc75 has been indicated, molecular bases underlying the conversion of a bacterial protein to the protein import channel during endosymbiosis are unknown. My group has been taking biochemical and molecular genetic approaches to address these unanswered questions. First, a polyglycine-strech was identified as a necessary signal for proper targeting of Toc75 to the chloroplast envelope. Further studies have suggested that the polyglycine may play a role in targeting multipleproteins to the chloroplast envelope. Second, we demonstrated that one of thylakoidal processing peptidases is involved in complete maturation of Toc75. This enzyme, which is located in both the envelope and thylakoids, now serves as a tool to examine the mechanism of thylakoid development. Finally, we showed in Arabidopsis that both Toc75 and its paralog, which appear to have diverged early in chloroplast evolution from an ancient cyanobacterium, are essential for embryogenesis. Defining the function of the Toc75 paralog should help us elucidate molecular bases of evolution of the protein import channel. Addressing specific questions about Toc75 has led us to various findings and provided tools to answer general questions about organelle biogenesis.
Supported by US-DOE Energy Biosciences and US-NSF Molecular and Cellular Biosciences Programs.
講師
Associate Professor, Department of Plant Sciences, University of California, Davis
井上 健太郎 博士
| 日時 | 2011年8月22日(月) 開場 15:30〜 開演 16:00〜(講演時間:60分) |
|---|---|
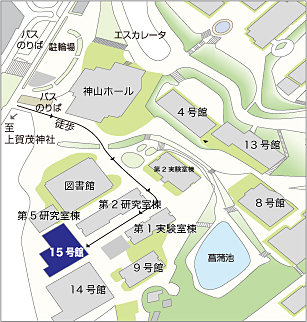
| 場所 | 京都産業大学総合生命科学部15号館 15102セミナー室 |
| 備考 | 英語講演(通訳無し)・事前申込不要・入場無料・一般の方の参加歓迎 |
| 主催 | 京都産業大学 総合生命科学部 |
問い合わせ
京都市北区上賀茂本山
京都産業大学総合生命科学部事務室
Tel.075-705-1466